
Voula Kanelis
-
E-mail:
-
Phone:
-
Fax:
-
Website:
-
Mailing Address:
3359 Mississauga Road
Mississauga ON L5L 1C6
Canada
Research Areas:
Biochemical and biophysical studies of ABC transporters and phage proteins
Research Profile:
Research in the laboratory is focused on two main areas: ABC transporters and phage proteins. We use a combination of NMR spectroscopy and other biophysical tools to obtain information about the structure, interactions, and dynamics of proteins.
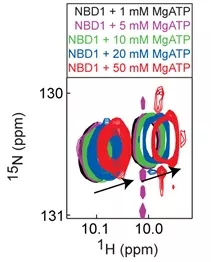
ABC transporters are multi-domain, integral membrane proteins found in all species and consist of two repeats each comprised of a membrane spanning domain (MSD) and a cytoplasmic nucleotide binding domain (NBD). ATP binding and hydrolysis at the NBDs results in transport of solutes across biological membranes. ABC proteins are of vast biomedical importance. ABC protein-mediated pumping of cytotoxic molecules from cells confers multidrug resistance to bacterial and tumour cells. Furthermore, many genetic diseases are caused by mutations of specific ABC proteins. We are studying two different ABC proteins in the lab: (1) Ycf1p, a Cd transporter in yeast; (2) sulfonylurea receptors (SUR), which form regulatory subunits in K+ channels. Studies of Ycf1p are aimed at understanding the basic mechanisms of regulation of ABC transporters. Studies of SUR proteins are aimed at understanding the molecular defects caused by disease-causing mutations in the proteins and how we can correct these defects with drugs.
Our work on phage proteins is focused on an HNH endonuclease a the lambda-like bacteriophage. The HNH protein is critical for the phage replication cycle. We are performing structural studies of the protein to understand the molecular basis for its function in phage morphogenesis. This work will ultimately lead to generation of designer phages that can be used to treat antibiotic resistant bacteria.
Courses Taught:
CHM361H5, CHM362H5, CHM373H5, and JCP463H5 (undergraduate); CHM1056H1 (graduate)
Publications
Recent:
Law WWH*, Kanelis V*, Zamble DB. Biochemical studies highlight determinants for metal selectivity in the Escherichia coli periplasmic solute binding protein NikA. Metallomics 2022, 14 (11), mfac084, https://doi.org/10.1093/mtomcs/mfac084 Impact factor: 4.6
Note that this work was started when WWH Law was in the laboratory of Professor DB Zamble. I assumed supervision of WWH Law, a PhD student in Chemistry, and the work after Professor Zamble unexpectedly passed away in July 2020.
Woloschuk, R. M., Reed, P. M. M., Jaikaran, A.S.I., Demmans, K.Z., Youn, J., Kanelis V. , Uppalapati, M., and Woolley, G.A.(2021) Structure-based design of a photoswitchable affibody scaffold, Protein Sci. 30, 2359-2372, 10.1002/pro.4196
Bickers, S.C., Benlekbir, S., Rubinstein, J.L. and Kanelis V. Structure of Ycf1p reveals the transmembrane domain TMD0 and the regulatory region of ABCC transporters. Proceedings of the National Academy of Sciences of the United States of America (PNAS). 2021 May 25,118(21):e2025853118. doi:10.1073/pnas.2025853118.
Weiditch SA, Bickers SC, Bona D, Maxwell KL, Kanelis V. HK97 gp74 Possesses an α-Helical Insertion in the ββα Fold That Affects Its Metal Binding, cos Site Digestion, and In Vivo Activities. J Bacteriol. 2020 Mar 26;202(8):e00644-19. doi:10.1128/JB.00644-19. Print 2020 Mar 26. PMID: 31988081
Bickers SC, Sayewich JS, Kanelis V. Intrinsically disordered regions regulate the activities of ATP binding cassette transporters. Biochim Biophys Acta Biomembr. 2020 Jun 1;1862(6):183202. doi: 10.1016/j.bbamem.2020.183202. Epub 2020 Jan 20. PMID: 31972165 Invited Review.
Weiditch S.A., Seraphim TV, Houry WA, Kanelis V. Strategies for purification of the bacteriophage HK97 small and large terminase subunits that yield pure and homogeneous samples that are functional. Protein Expr Purif. 2019 Apr 4;160:45-55. doi:10.1016/j.pep.2019.03.017.
Sooklal CR, López-Alonso JP, Papp N, Kanelis V. Phosphorylation Alters the Residual Structure and Interactions of the Regulatory L1 Linker Connecting NBD1 to the Membrane-Bound Domain in SUR2B. Biochemistry. 2018 Nov 6;57(44):6278-6292. doi:10.1021/acs.biochem.8b00503.
Zhao, J., Beyrakhova, K., Liu, Y., Alvarez, C.P., Bueler, S., Xu, L., Caishung, X., Boniecki, M., Kanelis, V., Luo, Z-Q., Cygler, M., and Rubinstein, J.L. Molecular basis for the binding and modulation of V-ATPase by a bacterial effector protein. PLoS Pathogens. 2017 Jun 1;13(6):e1006394. doi: 10.1371/journal.ppat.1006394.
Alvarez, C.P., Stagljar, M., and Kanelis, V. Hyperinsulinism-causing mutations cause multiple molecular defects in SUR1 NBD1, 2017 Apr 25. doi:10.1021/acs.biochem.6b00681.
Kanelis, V. From ions to insulin. eLife.25159
De Araujo E.D., Alvarez C.P, López-Alonso J.P., Sooklal C.R., Stagjlar M, Kanelis, V. Phosphorylation- dependent changes in nucleotide binding, conformation, and dynamics of the first nucleotide binding domain (NBD1) of the sulfonylurea receptor 2B (SUR2B), Journal of Biological Chemistry, 290(37):22699-714 (2015).
