Our overarching research focus is the relationship between adult neural plasticity and social status. We use naked mole-rats to study this question because they exhibit the most rigidly organized social and reproductive hierarchy among mammals. Naked mole-rats are eusocial, which is essentially an extreme form of co-operative breeding; they live in large colonies of up to 300 individuals in which reproduction is restricted to a single breeding female (called the queen) and one to three breeding males. Importantly, breeders of both sexes are socially dominant over all other members of the colony; the remaining members of the colony, called subordinates, are kept non-reproductive by the presence of the queen. However, this reproductive suppression is not permanent: adult subordinates can become breeders if they are removed from the colony and paired with an opposite sex mate. Given that these animals are remarkably long-lived (up to 30 years in captivity), this reveals an exceptional sustained potential for plasticity mediated by the social environment.
Our current research program can be described as three inter-related lines of research, which ultimately come together to highlight the importance of social environment in shaping brain and behaviour in adulthood. First and foremost, we study the neuroendocrine mechanisms associated with social status. Second, we are interested in individual differences in social phenotype. Third, we investigate how status or changes in the social environment regulate adult neurogenesis.
Neuroendocrine control of social status and reproductive suppression
We have demonstrated that a transition in social status triggers morphological and functional changes in the brains of naked mole-rats (Holmes et al., PNAS, 2007). These changes occurred in brain regions that coordinate reproduction and reproductive behaviours in mammals.
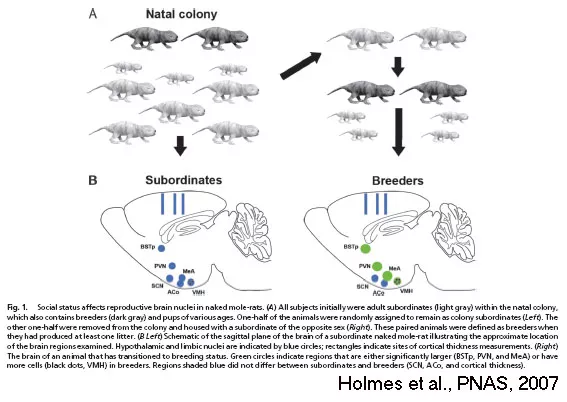
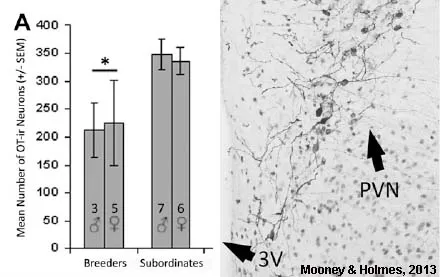
All of the brain regions that show morphological changes based on status are related to social behaviours. Thus, much of our work to date has explored a role for the hormone oxytocin, a small peptide that is important for social interactions and reproduction in diverse vertebrate species. We have found that subordinates have more oxytocinergic neurons than breeders in the PVN (Mooney & Holmes, Neuroscience, 2013), that status mediates expression of the oxytocin receptor in the MeA (Mooney et al., Neuroscience, 2015), and that peripheral administration of oxytocin increases prosocial behaviours (Mooney et al., Hormones & Behavior, 2014). Collectively these data indicate that oxytocin is a key player in promoting prosocial behaviour in subordinates.
Individual differences in social phenotype
Within the subordinate caste, further social stratification and divisions of labor exist: ‘soldiers’ (often larger, older subordinates) are observed to be more aggressive and defend the colony, whereas ‘workers’ (often younger, smaller individuals) participate in cooperative behaviors such as foraging and colony maintenance. While we are not the first to suggest subcastes within naked mole-rat subordinates, we have expanded on this early work to further characterize different subgroups of subordinates, to test whether these roles represent long term social phenotypes and to identify the neural mechanisms that underlie them (similar to our breeder/subordinate comparisons thus far). We have recently demonstrated that these subcaste classifications (based on behavioral specializations) remain stable in the long term but this stable task specialization becomes plastic when colony demands are altered (Mooney et al., Animal Behaviour, 2015). Removing work-, pup care-, and defense-specialists, caused remaining animals to fill open behavioural niches, demonstrating need-based task switching in eusocial mammals.
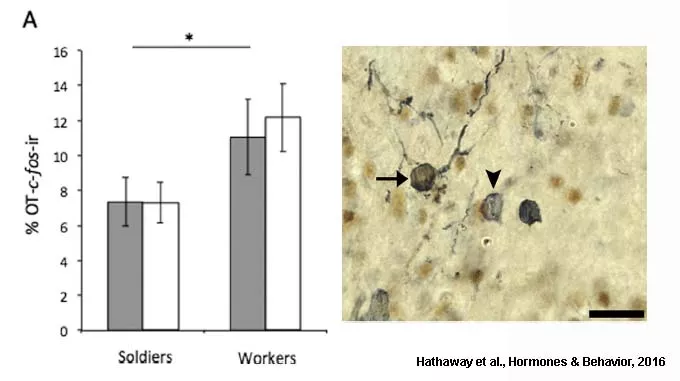
To begin to understand if oxytocin is also involved in subcaste differences in social behaviour, we tested whether activation of oxytocin neurons is associated with the distinct behavioural roles found in workers and soldiers. We found that subcaste differences exist between workers and soldiers for both behaviour and neural activation. Specifically, workers were significantly less aggressive than soldiers and showed greater activation of oxytocin neurons in the PVN when exposed to a novel conspecific (Hathaway et al., Hormones & Behavior, 2016).
Social control of adult neurogenesis
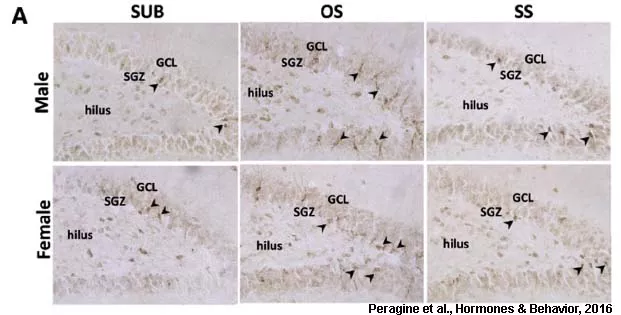
In mammals (including humans), some regions of the brain retain the ability to produce new neurons throughout life, a process called adult neurogenesis. The goal of this component of our research is to determine how social status controls neurogenesis in the mammalian brain and how these new neurons might contribute to status differences in behaviour. We found that breeders have significantly decreased neurogenesis in the dentate gyrus of the hippocampus compared to subordinates of either sex and this is particularly driven by low neurogenesis in the queens (Peragine et al., Neuroscience, 2014). We followed up on this by removing subordinates from their home colony and pairing them with either an opposite-sex or same-sex animal for one month and recorded behaviour of the pairs. Interestingly, we found the opposite-sex paired animals showed increased neurogenesis relative to subordinate controls while same-sex paired animals did not (though same-sex paired animals showed increased aggressive behaviour; Peragine et al., Hormones & Behavior, 2016). Collectively, these data suggest that neurogenesis is highly fluid as animals transition in status, likely based on the specifics of their social and endocrine environment.